|
This is why people being vaccinated against COVID-19 will be asked to stay at the vaccination site for a period of time following their injection, to ensure that anyone experiencing an allergic reaction can receive prompt treatment. Healthcare workers administering COVID-19 vaccines should be trained to recognise and treat serious allergic reactions. In some cases, precautions will need to be taken for people who have known allergies to previous doses of the vaccine or known components of the vaccine. If you have a history of allergic reactions, talk to your healthcare provider before you get vaccinated. A severe allergic reaction – such as anaphylaxis – is a very rare side effect of any vaccine. In very rare cases, some people may experience an allergic reaction after being vaccinated against COVID-19. 
Vaccines are continually monitored for as long as they are in use to detect and respond to rare adverse events. If you experience difficulty breathing, chest pain, confusion, loss of speech or mobility after your vaccine, contact your healthcare provider immediately. More serious or long-lasting side effects to COVID-19 vaccines are possible but extremely rare. If you are worried that the side effects that you are experiencing are unusual, if the pain in the arm where you got the injection gets worse after 24 hours or your side effects don’t go away in a few days, contact your healthcare provider for advice. You can manage any side effects with rest, plenty of non-alcoholic liquids and taking medication to manage pain and fever, if needed. Most side effects go away within a few days on their own. 
Not everyone will experience side effects. Side effects to COVID-19 vaccines include a fever, tiredness, headache, muscle ache, chills, diarrhoea and pain or redness at the injection site. This is a normal sign that the body is developing protection. Like with any vaccine, some people will experience mild to moderate side effects after being vaccinated against COVID-19. This is why vaccines are so closely monitored – to ensure that any issues with their production, storage or use can be rapidly identified and resolved. In this case, people who have received a vaccine from that batch may need to be vaccinated again to ensure they are protected. Recalls are generally associated with problems identified during the monitoring of batches of vaccines through quality control systems, stability studies and reports from the field, including cold chain issues where some vaccines have not been stored at the right conditions and so are no longer safe or effective. Vaccine recalls or withdrawals due to safety issues are rare. WHO works closely with vaccine manufacturers, health officials, researchers, and other partners to monitor any safety concerns and potential side effects. Investigations will take place to determine what exactly caused the event, and corrective measures will be put in place. In rare situations where a serious adverse reaction is suspected to be related to the vaccine itself, the vaccine may be temporarily suspended from use while the situation is assessed. GACVS is an independent group of experts providing authoritative, scientific advice to WHO on vaccine safety issues of global or regional concern. 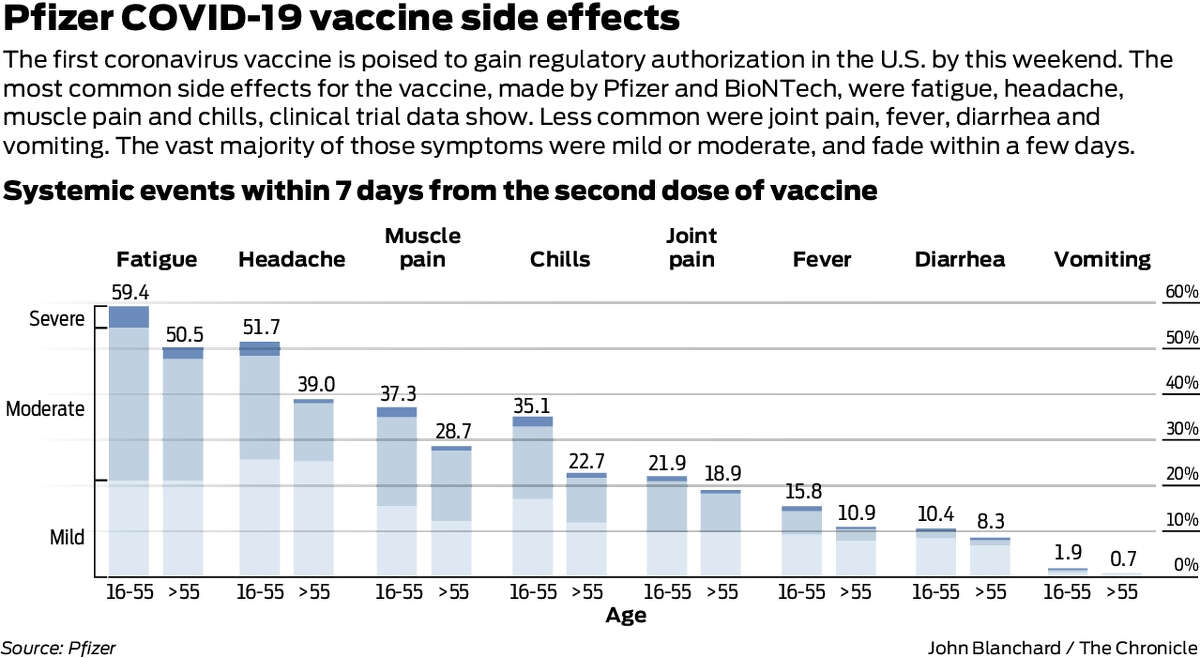
The WHO’s Global Advisory Committee on Vaccine Safety (GACVS) monitors how approved vaccines behave in the real world and to identify any signals of adverse events following immunization. In addition to reviewing data for regulatory purposes, the evidence must also be reviewed for the purpose of policy recommendations on how the vaccines should be used. 
SAGE then recommends whether and how the vaccines should be used. Independent reviews of the efficacy and safety evidence are required by WHO for each vaccine candidate, including regulatory review and approval in the country where the vaccine is manufactured, before WHO considers it for emergency use listing.Īn external panel of experts convened by WHO (the Strategic Advisory Group of Experts on Immunization (SAGE), analyses the results from clinical trials, along with evidence on the disease, age groups affected, risk factors for disease, programmatic use and other information. Some COVID-19 vaccine candidates have already completed their Phase III trials and are being used globally, and many other vaccine candidates are being developed and are still in the trial phase. The vaccines must be proven safe and effective in large Phase III clinical trials.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
